INTRODUCTION
Intensifying agriculture in Ukraine relies heavily on the proper use of mineral fertilizers (Nakaz Ministerstva 2021). Improper use, and gross violations of storage and transport conditions have led to the contamination of surface and ground waters throughout the river network. Only 5–10% of those applied are taken up by plants, and 90–95% are dispersed in the wind or washed out by rain and meltwater, entering streams, rivers, lakes, ponds and irrigation channels (Kutsenko 2004, Camargo et al. 2005, Batyan et al. 2009, Savci 2012, Dudnyk & Yevtushenko 2013, Henry et al. 2013, Stadnychenko 2013, Martynova et al. 2017, Karapetyants & Drakin 2018, Singh & Craswell 2021, Uvayeva et al. 2022a).
The usage of fertilizers is determined mainly by the character of different regions, in Ukraine divided into four: Polissia, Forest-Steppe, Steppe and Carpathian Mountains (Logvinov & Shcherban 1984). Each has characteristic soil types, water regime and precipitation. Polissia, northern Ukraine, has favourable precipitation but naturally nutrient-poor soils. This makes the use of nitrate fertilizers particularly effective. In Forest Steppe, centrally placed in Ukraine, regions to the west (right bank) of the Dnipro river, humid conditions and soil type favour the use of potassium fertilizers, while to the east (left bank), drier conditions favour the use of phosphorus fertilizers, as does the even more arid condition of the Steppe, where droughts are frequent (Pysarenko et al. 2008).
When transferred to water bodies, concentrations of nitrogen (as ammonium), potassium and phosphate ions increase. While not toxic themselves, they can change the direction of redox processes (Camargo et al. 2005, Dudnyk & Yevtushenko 2013). In alkaline environments, ammonium ions transform into ammoniac, highly toxic to aquatic animals (Metelev et al. 1971). It can induce convulsions, and blocks respiratory pathways, leading to mass mortality in both fish and molluscs. 90% or more of ammonium nitrate is transformed, and in carp ponds pH reaches 7.5–9.0. Phosphates can also cause problems, as when natural apatites are used, heavy metal contamination can occur, most especially in fish ponds (Dudnyk & Yevtushenko 2013, Singh & Craswell 2021).
These mineral fertilizers are often used in concentrations above those allowed by law, not only in agriculture, but also in fisheries (Solomatina et al. 2019), which can worsen water quality, sometimes resulting in hypoxia. Hence, there is a need to study the mode, direction and level of these accidentally polluting effects, especially on the molluscan fauna. It is known (Romanenko 2001, Kremser & Schnug 2002, Savci 2012) that at certain concentrations they cause pH and oxidation level to increase, along with mineralisation and eutrophication. Photosynthetic blooms result in accumulation of undecomposed detritus, leading to anoxic conditions. Excess of phosphates also disturbs the calcium balance in molluscs (shell thinning) (Batyan et al. 2009, Pimenova 2011). Excess of nitrates leads to oxygen starvation in molluscs, decrease of hemolymph Hb concentration, and weakening of heart and vessel muscles (Martynova et al. 2017, Karapetyants & Drakin 2018). Excessive use of potassium fertilizers threatens the molluscs with dehydration due to excessive osmotic loses (Radkevich 1998, Chernova & Bilova 2004).
Planorbid snails are suitable to examine some of these effects. They have two methods of respiration, with the lungs, and by direct absorption of oxygen through the body surface (Régondaud 1961). In pulmonary respiration, air is taken into the lung via a long moveable siphon. In normal conditions, both methods are of similar capacity, 0.025 (lung) and 0.03 (surface) mg O2/hour per 1 g of fresh mass, respectively (Prosser & Braun 1967, Stadnychenko 2013). This study sets out to establish the effects of different concentrations of nitrate, potassium and phosphate fertilizers on the respiration, in both modes, of the great ramshorn snail Planorbarius corneus (Linnaeus, 1758), taking separately its two vicariant allospecies, “eastern” and “western” (Mezhzherin et al. 2005). These allospecies differ from each other by conchological, anatomical, chorological and ecological features (Garbar 2003, 2009, Garbar & Garbar 2006, Garbar et al. 2020, Babych et al. 2021, Uvayeva et al. 2022b). The species is abundant and widespread in both running and still waters. Our material comes from water bodies in the Polissia and Forest-Steppe zones in 2020–21; material from the Steppe zone, due to be sampled in 2022, has become unavailable to us for reasons beyond our control.
MATERIAL AND METHODS
Mature individuals of the great ramshorn P. corneus s. lato (Linnaeus, 1758) allospecies (Figs 1–6), collected by hand in June–July 2020–2021 in four localities (Fig. 7). “Western” allospecies (diameter of shell 25.84±0.09 mm): 277 ind. from Teteriv River (Zhytomyr, Zhytomyr region; 50°14'19.5"N, 28°40'49.8"E), and 235 from Hnyla River (Horodnytsia village, Ternopil region; 49°24'38.4"N, 26°00'31.9"E), and “eastern” allospecies (diameter of shell 22.63±0.12 mm): 293 ind. from Snov River (Snovsk, Chernihiv region; 51°49'15.9"N, 31°55'27.5"E), and 294 from Psel River (Sumy, Sumy region; 50°55'20.3"N, 34°49'30.1"E) were used for studying the pulmonary and direct diffusive respiration, respectively (Figs 8–11).
Figs 1–6
Shells of Planorbarius corneus s. lato: 1–3 – allospecies “western” (Teteriv river, Zhytomyr); 4–6 – allospecies “eastern” (Snov river, Snovsk); 1, 4 – top view; 2, 5 – bottom view; 3, 6 – side view. Scale bars 10 mm

Fig. 7
Map showing the localities of Planorbarius corneus s. lato allospecies: I – Polissia (black circle – Teteriv river, Zhytomyr; black squares – Snov river, Snovsk); II – Forest-Steppe (black circle – Hnyla river, Horodnytsia village; black squares – Psel river, Sumy); III – Steppe; IV – Carpathian mountain region; black circle stand for “western” and black squares for “eastern” allospecies
Figs 8–11
Habitats of great ramshorn Planorbarius corneus s. lato: 8 – Teteriv River (Zhytomyr, Zhytomyr region); 9 – Hnyla River (Horodnytsia village, Ternopil region); 10 – Snov River (Snovsk, Chernihiv region); 11 – Psel River (Sumy, Sumy region); 8–9 – allospecies “western”; 10–11 – allospecies “eastern”

Allospecies were identified by their conchological features (Garbar 2003, Mezhzherin et al. 2005). Before the toxicology experiment, the selected individuals were acclimatised over 15 days in laboratory conditions (Khlebovich 1981): 10 l water volume, 4 ind/l density, 20–22 °C temperature, 7.6–8.3 pH, 8.4–8.9 mg/L O2 oxygenation. Renewing of conditions, and feeding were daily, and the snails were fed on Cladophora sp. and Myriophyllum spicatum L., obtained from the water bodies from where the snails originated.
The snails were weighed using electronic scales, WPS 1200/cm, and their shells were measured using calipers. The volume of hemolymph was measured after the experiments by insulin syringe via complete bleeding of the animals. Hb content was measured following the Sali method with the Alyakrinskaya (1970) modification, and hemolymph pH – by indicator strips pH-TEST (production of PRC). The snails were fed daily with Myriophyllum sp. in the experimental arena.
The main toxicology experiment was conducted following Alekseyev (1981). Toxicants (p.a.) – NaNO3, K2SO4, Ca(H2PO4)2, – were used in concentrations indicated in MPC: 0.5, 1, 2, 3. The value of the maximum permissible concentration (MPC: mg/L) for great ramshorn is for NaNO3 – 0.01, K2SO4 – 0.005, Ca(H2PO4)2 – 0.001. Their solutions were prepared with aged (2 days) tap water. Duration of experiment – 7 days. The indexes of pulmonary and direct diffusive respiration were estimated by observation of behavioral and physiological reactions of experimental individuals to the applied mineral fertilizers (Stadnychenko et al. 1996). The number of inspirations was calculated as number of emerging events through the water surface tension film with pneumostome mounting to its bottom surface. The successful perforation was usually followed by a loud clap. It signified the beginning of inspiration. The duration of inspiration was considered as the time from the clap to the snail detaching from the water surface tension film and sinking to the bottom. The volume of inspiration was estimated by the number of bubbles appearing from the lung of each snail after they had been given some mechanical irritation, usually by being pricked by a pin repeatedly until no more bubbles emerged.
The level of diffusive respiration was estimated by the duration of presence of living signs in experimental animals, which were deprived of access to pulmonary respiration. For this, they were placed at the aquarium bottom into the perforated (water-permeable) plastic boxes (7 × 7 × 7 cm) with mounted weight.
The package “STATISTICA 6.0” (Borovikov 2013) was used to analyze the data obtained from the experiments.
RESULTS
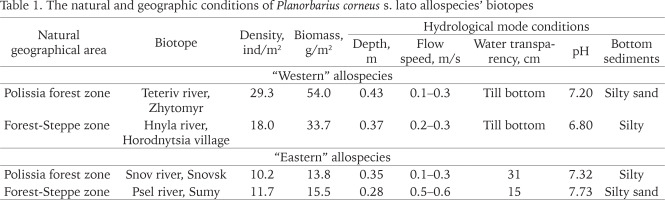
The samples of great ramshorn snail P. corneus s. lato were collected in the water bodies of two Ukrainian nature-geographical zones – Polissia and Forest-Steppe, divided by Dnipro River into Right and Left Banks. “Western” allospecies inhabits Right Bank of both zones, while “eastern” one is widespread on the Left Bank of both (Figs 1–6). The information on natural and geographic conditions of their ranges is presented in Table 1.
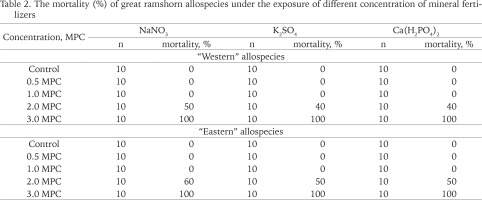
Table 2 provides the data on the different levels of sensitivity and durability to the impact of mineral fertilizers of three types in the two P. corneus allospecies. Mortality was higher among the “eastern” allospecies experimental individuals under all treatments in all applied concentrations (Table 2).
Table 2
The mortality (%) of great ramshorn allospecies under the exposure of different concentration of mineral fertilizers
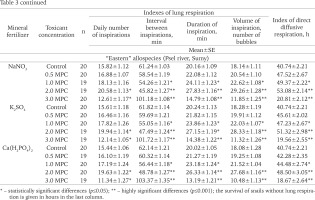
The effects of different mineral fertilizers on the pulmonary and surface diffusive respirations of P. corneus allospecies are given in Table 3. With toxicant concentrations rising from 0.5 MPC to 2 MPC the snails showed an increase of daily inspiration number, duration and volume along with shortening of intervals between them. At 3 MPC of the same toxicants, however, the snails showed a decline in indexes of pulmonary respiration with longer intervals between them. At 3 MPC, survival in the absence of pulmonary respiration was always shorter than in the controls, but it was sometimes more prolonged than in the controls at lower concentrations.
Table 3
The impact of mineral fertilizers in different concentrations on indexes of lung and direct diffusive respiration of Planorbarius corneus s. lato allospecies
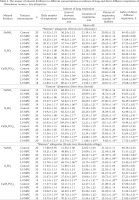
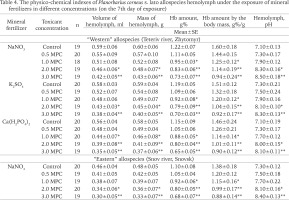
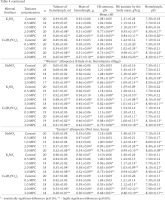
Table 4 shows the significant differences between the allospecies by some indexes of their internal medium (hemolymph) when treated with the different mineral fertilizers in concentrations from 0.5 to 3 MPC. Experimental snails showed a decrease in hemolymph volume, mass and Hb content, and an increase in pH.
DISCUSSION
The ionic components of these fertilizers, nitrates, sulphates and phosphates, enter the snails percutaneously (Dudnyk & Yevtushenko 2013, Henry et al. 2013, Singh & Craswell 2021). They accumulate in the hepatopancreas, but are widely distributed around the body by the haemolymph. They can disturb the natural balance of the metallic ions essential for normal function (Batyan et al. 2009, Henry et al. 2013, Singh & Craswell 2021, Presa et al. 2022).
Our experiments establish the limiting concentrations of the toxicants involved, in terms of their effects. In absolute terms, nitrates were the most toxic, and phosphates the least. For pulmonary respiration, the pattern is one of increasing respiration rates up to 2 MPC, presumably a response to stress; number of intakes, their frequency, duration and volume all increase. At 3 MPC, however, there are clear signs of systemic collapse; respiration is severely curtailed. A similar effect at 3 MPC is seen in survival time when only subcutaneous respiration is possible, although there is an anomalous increase in survival time at lower concentrations relative to controls, perhaps indicating a decrease in metabolic activity. Some clues as to the mechanism of interference in normal respiratory processes are given by the changes in hemolymph volume, composition and pH. The toxic effects include both the reduction in volume and a decrease even in the relative amount of Hb present. All these effects increase markedly at 2 MPC and above.
While the changes in respiration and survival in response to increasing concentrations of pollutants followed the same pattern in both “western” and “eastern” allospecies, the “eastern” is, on all measures, more vulnerable than the “western”. The “western” form is found in the western and central part of Right Bank Ukraine, while the “eastern” is found to the east and north-east of the Left Bank, referring to the Dnipro River. It also occurs in the extreme south of Ukraine. While it is unclear why such a difference should exist, we note that the “eastern” form lives in areas generally more arid than those occupied by the “western” form (Harbar et al. 2021). The use of species like the great ramshorn snail as bioindicators of pollution requires a detailed knowledge of such variation in tolerance.